|
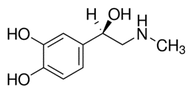
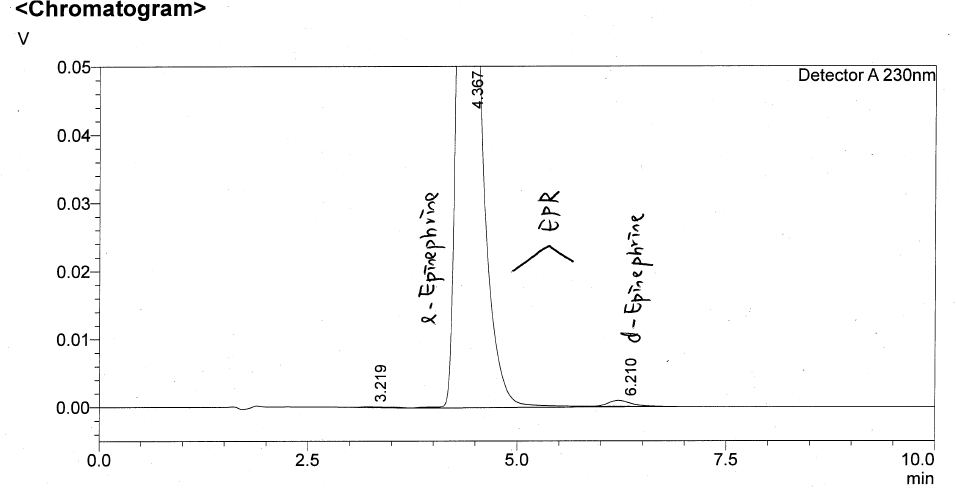
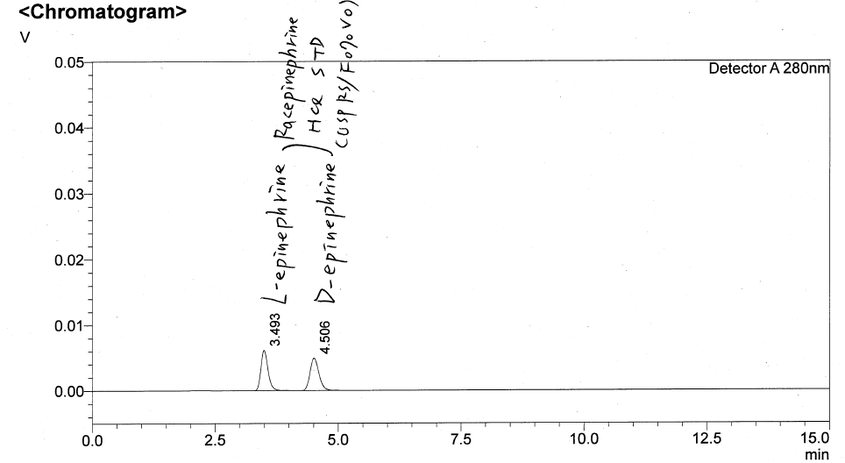
Enantiomeric purity in chiral molecules – such as Transo-Pharm’ s Epinephrine Base and Bitartrate – have been in the focus of the health authorities for some time now. Transo-Pharm is well prepared to fully satisfy the more stringent requirements with their state-of-the-art stereo-selective manufacturing process. Transo-Pharm is therefore in the position to achieve an Enantiomeric Excess (ee) of 99% in commercial production for several years now. This is a significant contribution to ensure patient safety. Please find the full article below. Learn more about the science behind this topic. Chirality is one of those fundamental properties of molecular systems which are ubiquitous in nature. Notably, a number of important biological compounds including proteins, amino acids, nucleosides, sugar, and a number of hormones, which are the basic building blocks of life, are chiral and thus the chemistry of many fundamental biological (metabolic and regulatory) processes are directly controlled by such molecular systems [1]. Chiral molecules are stereoselective with regard to specific biological functions. Enantiomers differ considerably in their physiological reactions with the human body [1]. Adrenaline/Epinephrine as part of Transo-Pharm´s product portfolio is such a typical example. Natural occurring Adrenaline emerges in R-configuration and turns the oscillation plane of light to the left ([α] 20/D: -50.0 to -54.0°). L-Adrenaline (R-(-) Adrenaline) R-Epinephrine is approx. 15 to 40 times more pharmacologically active than its S- enantiomer (D-Adrenaline). Therefore, high purity of the “right” enantiomer has to be ensured by the production process and efficiently analytically controlled. Patient safety has to be safeguarded! Asymmetric synthesis is a mature approach to the production of single enantiomers [1] compared to the old-school racemate segregation by means of diastereomer precipitation. Since Transo-Pharm well established a stereo-selective process we have been in the position to supply our customers with commercial L-Adrenaline showing an Enantiomeric Excess (ee) of 99% for some years now. The applied homogenous catalyst is not commercially available in the market. Therefore, it is manufactured in-house in Germany using our own IP, which ensures a reliable & robust supply chain. Illustrative ee – inhouse HPLC chromatogram Illustrative ee – USP HPLC chromatogram (SST) Conclusion Transo-Pharm´s homogenous enantioselective Epinephrine synthesis stands out due to:
0 Comments
Your comment will be posted after it is approved.
Leave a Reply. |
Categories
All
Archives
June 2024
|
||||||
ServicesProducts |
Company |
|

 RSS Feed
RSS Feed

